Precise Control Over Dissolved Oxygen (DO) Levels
Simulate Real Oxygen Microenvironments
Dissolved oxygen (DO) is a critical factor in cell biology, influencing processes such as energy metabolism, gene expression, and cell signaling. Whether you’re studying cells under hypoxic conditions, mimicking normoxia, or supplying additional oxygen to your system, precise control over oxygen levels is essential — yet difficult to achieve with traditional methods like hypoxic chambers, gas purging, or chemical scavengers.
Our custom-built electrochemical systems offer a powerful alternative: finely tunable, electrochemically generated oxygen gradients that can be spatially localized and dynamically modulated in real time. With well-characterized electrode materials and programmable protocols, you can reliably simulate hypoxia, anoxia, or physoxia directly in your culture system — and adjust conditions within seconds, not hours.
Why Choose Electrochemical Oxygen Control?
Traditional Approaches Have Limitations
Hypoxia chambers and gas-perfused systems are slow, coarse, and global in their effects. They expose entire cultures to uniform oxygen conditions and often require hours to equilibrate. This makes it difficult — if not impossible — to study fast, localized, or dynamic oxygen effects, particularly in high-throughput or live-cell imaging formats.
With our custom-built electrochemical systems, you can:
- Create stable or dynamic oxygen gradients
- Switch oxygen levels in seconds
- Mimic real physiological scenarios
- Run parallelized experiments with multiple oxygen profiles simultaneously
How It Works
At the core of our oxygen control system is the four-electron oxygen reduction reaction (ORR) on platinum cathodes — a highly efficient and selective process that consumes dissolved oxygen and converts it into water [1] [2]:
O₂ + 4 H⁺ + 4 e⁻ → 2 H₂O
This reaction, used in water deaeration systems [3-8], is now being adapted for life science applications [9] [10].
To increase oxygen levels, we use Oxygen Evolution Reaction (OER). By applying a controlled potential at the anode, we trigger the following reaction:
2 H₂O → O₂ + 4 H⁺ + 4 e⁻
This approach enables precise oxygen enrichment directly within the culture system. Recent studies confirm its safe and stable performance in physiological environments, with minimal pH drift and no harmful byproducts [11].
By adjusting electrochemical protocol, electrode configuration and well architecture we can precisely control both the depth and shape of oxygen gradients — at spatial scales ranging from microns to full-well coverage. Since oxygen is naturally replenished by diffusion, both steady-state and oscillating conditions can be achieved.3
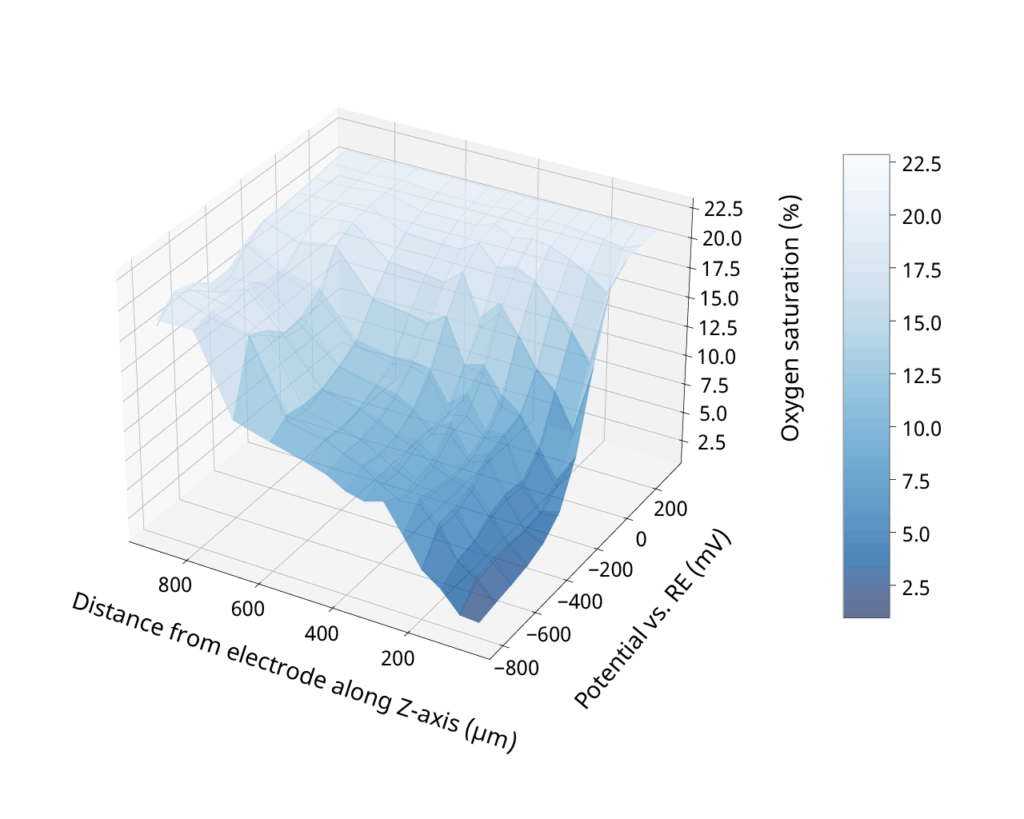
Oxygen gradient induced electrochemically as a function of electrode potential and distance from the electrode surface.
Ready to Simulate Real-World Oxygen Microenvironments?
Book a call with us to explore how our custom electrochemical systems can enable your oxygen-dependent experiments.
References
[1] Ehlich J, Vašíček Č, Dobeš J, Ruggiero A, Vejvodová M, Głowacki ED. Shattering the Water Window: Comprehensive Mapping of Faradaic Reactions on Bioelectronics Electrodes. ACS Appl Mater Interfaces. 2024 Oct 9;16(40):53567-53576. doi: 10.1021/acsami.4c12268.
[2] Ehlich J, Migliaccio L, Sahalianov I, Nikić M, Brodský J, Gablech I, Vu XT, Ingebrandt S, Głowacki ED. Direct measurement of oxygen reduction reactions at neurostimulation electrodes. J Neural Eng. 2022 Jun 27;19(3). doi: 10.1088/1741-2552/ac77c0. PMID: 35688124.
[3] Holubowitch, Nicolas E.. “Membrane-free electrochemical deoxygenation of aqueous solutions using symmetric activated carbon electrodes in flow-through cells.” Electrochimica Acta (2018): n. pag.
[4] Utsav R. Dotel, Kai Vuorilehto, Magne O. Sydnes, Hans Urkedal, and Tor Hemmingsen. Electrochemical Oxygen Removal from Seawater in Industrial Scale Using Silver Cathode. Industrial & Engineering Chemistry Research 2017 56 (31), 8954-8960 DOI: 10.1021/acs.iecr.7b02004
[5] TAMMINEN , A., VUORILEHTO , K. Application of a three-dimensional ion-exchange electrolyte in the deoxygenation of low-conductivity water. Journal of Applied Electrochemistry 27, 1095–1099 (1997). https://doi.org/10.1023/A:1018446912269
[6] Vuorilehto, K., Tamminen, A. & Ylasaari, S. Electrochemical removal of dissolved oxygen from water. J Appl Electrochem 25, 973–977 (1995). https://doi.org/10.1007/BF00241593
[7] Gurskii, V.S., Kirpikov, D.A., Kharitonova, E.Y. et al. Catalytic deoxygenation of high-purity water using membrane electrode units. Russ J Appl Chem 88, 1656–1660 (2015). https://doi.org/10.1134/S107042721510016X
[8] Marei MM, Roussel TJ, Keynton RS, Baldwin RP. Electrochemical dissolved oxygen removal from microfluidic streams for LOC sample pretreatment. Anal Chem. 2014 Sep 2;86(17):8541-6. doi: 10.1021/ac501398f.
[9] Wong JJY, Varga BV, Káradóttir RT, Hall EAH. Electrochemically induced in vitro focal hypoxia in human neurons. Front Cell Dev Biol. 2022 Sep 28;10:968341. doi: 10.3389/fcell.2022.968341.
[10] Hill MH, Meloni GN, Frenguelli BG, Unwin PR. Transient Single Cell Hypoxia Induced by Localized Galvanostatic Oxygen Challenge. ACS Meas Sci Au. 2025 Apr 1;5(2):234-241. doi: 10.1021/acsmeasuresciau.4c00100.
[11] Lee I, Surendran A, Fleury S, Gimino I, Curtiss A, Fell C, Shiwarski DJ, Refy O, Rothrock B, Jo S, Schwartzkopff T, Mehta AS, Wang Y, Sipe A, John S, Ji X, Nikiforidis G, Feinberg AW, Hester J, Weber DJ, Veiseh O, Rivnay J, Cohen-Karni T. Electrocatalytic on-site oxygenation for transplanted cell-based-therapies. Nat Commun. 2023 Nov 9;14(1):7019. doi: 10.1038/s41467-023-42697-2.